
Industrial Design Trends: What Medical Device R&D Leaders Need to Know
- Posted by Ryan Zumbach
- On October 2, 2019
- human factors, Industrial design, usability
Medical devices have particular industrial design needs that set them apart from consumer products. In this article, we explore what makes medical devices unique, and provide a summary of recent industrial design trends: aesthetic, style, color, material, and finishes applicable to the needs of medical devices. We are sharing our experience based upon designing medical devices and similar systems. At the end of this article, we provide actionable guidance that medical device R&D leaders should keep front of mind before commissioning design projects!
INDUSTRIAL DESIGN DEFINED
Industrial Designers are studied professionals who create the look and plan the function of products; balancing usability, aesthetics, and pragmatic considerations. Said another way, industrial designers are like architects, creating products rather than buildings (concept credit to IDSA).
Architects don’t realize buildings by themselves, they collaborate with the building trades, landscapers, inspectors, and others. In the same way, Industrial Designers collaborate with marketers, engineers, manufacturing professionals, and many others to realize new products. Mixing art and science, Industrial Designers are the renaissance men and women who tie it all together upfront. If you truly love your phone, watch, car, mixer, or hand tool, an Industrial Designer likely had something to do with it.
WHY INDUSTRIAL DESIGN AND FORM, STYLING, AND CMF MATTER IN MEDICAL DEVICES
Medical devices are used in life-critical situations where injury or death could result from product failure or misuse. As a result, human factors, usability engineering, and aesthetic optimization are of utmost importance in medical devices. Medical devices are used in stressful situations where life is on the line, and a poorly-placed button or confusing indicator could make a real life-or-death difference.
In addition, the US and International regulations require the use of human factors and usability engineering in medical device design. Not only does good industrial design make better devices, but it is also required to legally market devices in many worldwide markets.
Thoughtful human factors, usability, and aesthetic considerations translate to better outcomes for the end-user – whether that person is a clinician, patient, or other stakeholders. Conducting user research and examining findings to define human factors, usability, and aesthetic requirements upfront in the design process leads to products that reduce use errors, minimize patient harm, lower clinician liability, and reduce product liability exposure.
Real-World Example of Successful Industrial Design Practice
To see a well-designed medical device, check out the Philips IntelliVue X3! This product is a portable patient monitor that streamlines clinician workflow by reducing steps in the preparation of patient transport while preventing gaps in patient monitoring. A great outcome complemented by thoughtful and well-executed industrial design.
The IntelliVue X3 is an adaptable and customizable solution that mimics the design metaphor of a smartphone. The screen auto-rotates to provide the best viewing angle and has intuitive touch gesture controls reminding users of operating their personal smartphones. The GUI (graphical user interface) uses large, color-coded text, customizable to show the most relevant information on command.
Strong design and aesthetic features were carefully considered, and the results produce a quick initial understanding of the product, even to clinicians unfamiliar with this specific monitor. The large handle is a good example of exaggerated affordance; integrated and easily recognizable enhances accessibility to the user. A contrasting black and white monochromatic theme make this interaction point even more visible at first glance. Clinicians comment on how “sleek” and “modern” the device looks – an interpretation due to the simple form and professional black and white color palette, design that elicits a feeling of performance and professionalism. Smooth surfaces and anti-microbial materials were selected to allow easy cleaning.

Many beneficial features make the Philips IntelliVue X3 a successful product. However, some medical devices miss the mark on human factors and aesthetics. If the aesthetics of a product are neglected, negative consequences on usability will result in major harm to patients, clinicians, and caregivers.
Real-World Example of an Unsuccessful Industrial Design Practice
A poorly-designed medical device can be confusing to operate, leading to user errors. One example that stands out is the EpiPen epinephrine auto-injector device by Mylan. It falls short in satisfying several human factors and usability engineering needs.

EpiPen has a design metaphor of a pen – the aesthetic analogy of a cap and needle to a cap and ballpoint. This is reinforced by the form of the device and further solidified by the brand name. However, this design metaphor is broken. Studies have shown both users and physicians are prone to using the device incorrectly which leads to unintentional injections. According to this study, between 1994 and 2007 there were 15,190 unintentional injections. Of all voluntarily reported unintentional injections, 76% occurred in the finger or thumb, followed by 14% in the hand or palm.
This signifies a major flaw with the design and aesthetics leading to harmful repercussions. Users are trained that removable caps provide access to functional elements – pens, water bottles, or USB flash drives.
- The EpiPen cap is assumed to protect the needle, but the cap is a safety release for the needle on the opposite end.
- Additionally, the cap is often misunderstood to be a button; an aesthetic issue resulting from the design.
- If users do figure out that they need to remove the cap, they are typically inclined to flip the device expecting the needle to come out of the cap end.
Three design strikes and you’re out – users often place their hands/thumbs on the needle resulting in unintentional injections.
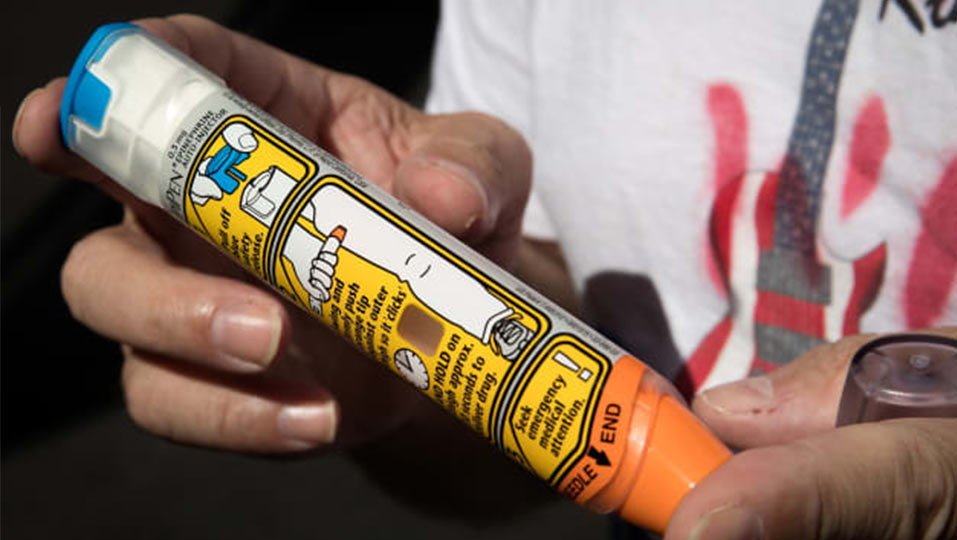
By uncovering user needs early in the design process, aesthetic considerations can be developed in tandem with the technology platform to avoid developing hazardous devices that lead to unintentional harm.
Engenious Design thinks broadly about how aesthetic considerations should be applied through form, styling, and CMF strategy. We consider a broad range of stakeholders in pursuit of inclusive design that performs for everyone. Ergonomic considerations such as body geometry data that inform suitable proportions and size of the device’s form. Engenious Design also incorporates insights for users’ age, cognitive abilities, physical health, and physical ability.
Alignment to user scenarios and our understanding of the medical use environment uncovers additional key needs, activities, and features that impact design decisions. All these help unlock user experiences – improving new user onboarding, reducing patient/clinician stress, and meeting mandated FDA requirements that allow distribution of medical devices in the US and EU.
Applying a human-centered approach brings a much-needed human element to technology in the medical device industry. Form, styling, and CMF strategy established by this background research and understanding improve medical device performance and increases the overall perception of quality. Allowing Engenious to create reliable and dependable experiences customers will prefer for years to come and lead to strong brand identity in the market.
HOW INDUSTRIAL DESIGN TRENDS FOR THE MEDICAL INDUSTRY VARIES FROM CONSUMER PRODUCTS
Design for medical devices requires special attention and diligence to mitigate hazards and user risks. A medical device’s form, styling, and CMF choices need to closely align with the design requirements. Contrasting with consumer products that arguably have more aesthetic freedom. Consumer products satisfy the most current needs and wants of a market or create a desire for products consumers did not previously know they want. The development timelines are rapid with low barriers to market entry; allowing consumer products to keep up with fads and quickly adopt contemporary industrial design trends or shift between trends to reach niche target markets. This structure allows for typically short product lifecycles; products may become obsolete quickly. In contrast to consumer products, medical devices demand more careful consideration of the product’s lifecycle.
Medical Devices Have a Long-Life Cycle; Design it to be Timeless!
It’s not uncommon for medical devices to have a 10-15-year useful field life; that’s long! As a result, it is imperative to plan the aesthetic styling and form language to stand the test of time and expected product lifecycle. For this reason, we are careful to not chase short-lived industrial design trends and aesthetic fads based on the expected field life – planning for a more timeless aesthetic that is likely to withstand a long lifecycle. It is important for a medical device to achieve an aesthetic that is perceived as the latest technological advancement – at market launch and a decade later. If the device is perceived by patients and clinicians to appear outdated before the technology becomes obsolete, users may become skeptical of the technology behind the device and question its reliability, and the brand of the device manufacturer. That’s not good!

Safety 1st Fever Light Ear Thermometer
For example, the Safety 1st Fever Light Ear Thermometer is a product on the market today that is aesthetically outdated. Reminiscent of the late ’90s to early 2000’s product in form, styling, and CMF. Its sweeping arcs and ellipses paired with dull colors don’t command a sense of trust that this device is reliable or contains up to date technology. Users associate the aesthetic of this device and its perceived performance with products of the past in the same way a modern Nest thermostat stands apart from a traditional home thermostat.
3 INDUSTRIAL DESIGN TRENDS TO APPLY TO MEDICAL DEVICES TODAY
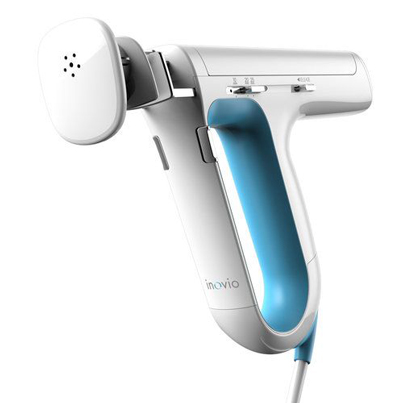
1) Exaggerated Affordances
An affordance is a universal visual cue that indicates how someone may interact with a product. Important user interface elements on a medical device draw attention through exaggeration of an affordance. Check out the large blue handle on the Inovio which represents this concept. Affordances are particularly helpful for healthcare professionals using an object for the first time when their hands are full, or their mind is preoccupied.

2) Organic and Approachable
Approachable and comforting in appearance, an organic aesthetic may be a great fit for certain classes of medical devices. Soft forms communicate comfort, visually tying to objects found in nature. The organic and approachable design aesthetic provides the opportunity to humanize objects in an environment that needs more personal appeal to overcome a traditionally sterile environment or to reduce patient anxiety common in clinical situations.

3) Black and White Monochromatic
Black and white monochromatic contrast is an aesthetic trend that is interpreted as modern, sophisticated, and timeless. These classic colors paired with brushed or chrome metal accents add details of luxury and professionalism. This CMF (Color, Material, Finish) strategy creates a perception of precision, which is extremely important for many medical devices and tools. Because many medical devices need to last in the field for several years, black and white is a classic aesthetic that will stand up to shifting trends and styles – from changes in consumer electronics to hospital interior design trends.
A CONSUMER TREND THAT SHOULD NOT APPLY TO MEDICAL DEVICES

Rose Gold CMF
Apple popularized this color and material with its release of a special edition Apple Watch, followed by the colorway offered with the iPhone 6s. From its roots in jewelry and its transformation into consumer electronics, rose gold can be found nearly everywhere. In 2016, Pantone even named “Rose Quartz” the color of the year. The color may represent tranquility, calming, balance, and compassion (emotions needed in the medical industry), but this trend has become so popular that it will inevitably fade into a fad. This CMF trend is not a suitable professional aesthetic for critical care medical devices because many medical devices need to last in the field for several years. When the rose gold fad fades, medical devices that go all-in with this aesthetic trend may not be taken seriously by clinicians and patients.
DROP US A LINE
Engenious Design keeps up on the latest Industrial design news, design awards, and style trends. This allows us to make up-to-date recommendations for clients. We help you navigate the trends that apply to medical devices while steering clear of those that won’t inspire confidence from healthcare professionals and patients.
Curious to learn more about Engenious Design? Drop us a line or even set-up a time to come visit.




